Honey Caviar, Fourme d’Ambert, Black Tea
The honey caviar is a great way to bring molecular gastronomy to your cheese plate, desserts or drinks. In this recipe we are pairing the acacia honey caviar with a delicious Fourme d’Ambert cheese and a small cup of black tea.
Did you know that black tea pairs perfectly with acacia honey and Fourme d’Ambert? According to the molecular profiling done by FoodPairing.com, these three ingredients have several molecular compounds in common which makes them a great combination.
The honey caviar is made using the basic spherification technique developed by molecular gastronomy Chef Ferran Adria.

Making spherification caviar is fun but it can be a draining process to make enough for a few cocktails or dishes one drop at a time using just a syringe or squeeze bottle. With the Caviar Maker you can now multiply the production by 96 times with no extra effort! You can purchase the Caviar Maker from our store.
Honey Caviar Ingredients
- 110 g (3.9 oz) acacia honey
- 90 g (3.2 oz) water (filtered water or with low calcium content)
- 1.6 g sodium alginate (0.8%)
Honey Syrup
- 2 oz acacia honey
- 1 oz water
Calcium Bath
- 500 g (18 oz) water
- 2.5 g calcium chloride
Other Ingredients
- 200 g Fourme d’Ambert cheese
- Back tea
Preparation
1- Prepare the honey mix for the caviar. Mix the sodium alginate in the water using an immersion blender until the sodium alginate is completely dissolved. If this is your first time doing this, be aware that this may take longer than expected.
2- Once the sodium alginate is dissolved, mix with the honey using the immersion blender. Let the mix rest in the fridge for 12 hours to eliminate the air bubbles created by the immersion blender.
3- Once the mix has no air bubbles remove it from the fridge until it reaches room temperature to reduce the viscosity of the mix.
4- Prepare the calcium bath in a bowl by dissolving the calcium chloride in the water.
5- Prepare the honey syrup by mixing with a spoon the honey with the water until completely dissolved.
6- You are now ready to start creating the caviar! Fill a syringe with the honey-alginate mixture and expel it drop by drop into the calcium bath. The syringe needs to be high enough (about 6 inches from the bath surface) for the drops to sink and to prevent the formation of a tail. Don’t go too high or the drops may break into smaller drops creating “baby” drops.
7- Leave the caviar "cooking" for about 1 minute in the calcium bath and then carefully remove it using a sieve. Then rinse it very gently with water to remove the calcium.
8- Once you have enough quantity of caviar, add a few spoons of honey syrup.
9- Consume immediately since the jellification process continues even after removing the caviar from the calcium bath and will eventually convert into a solid gel sphere with no magical liquid inside.
Assemble and Serve
1- Place a few spoons of caviar with honey syrup on top of the Fourme d’Ambert cheese.
2- Serve with crackers and a small cup of black tea.





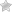 (6 votes, average: 3.33)
(6 votes, average: 3.33)
